|
P. gingivalis, one of the major etiological agents of periodontal disease, elicited complex changes when incubated with GECs: it suppressed the expression of histone deacetylase 1 and 2 (HDAC1 and HDAC2) and DNA methyltransferase 1 (DNMT1). Similarly to their pathogenic counterparts, nonpathogenic bacteria may also interact with GECs, modulating native immune responses to invading pathogens. In addition to Porphyromonas gingivalis, Tannerella forsythia, Treponema denticola, and Prevotella intermedia, the pathogenic bacteria causing periodontitis, a series of nonpathogenic bacteria (e.g., Fusobacterium nucleatum) also inhabit the oral cavity of periodontitis patients, forming a microbial community (reviewed by Ref. The cells of the innate immune system, including gingival epithelial cells (GECs), recognize pathogen-associated molecular patterns interacting with toll-like receptors (TLRs) and other receptor classes and respond by the production of antimicrobial peptides, such as human β-defensins and chemokines that activate adaptive immune responses as well. A critical step in periodontal disease progression is the disruption of the host innate immune system by anaerobic bacteria (reviewed by Ref. In addition to its local consequences (occasional pain, impaired mastication, loosening, and loss of teeth), subgingival infection may have a systemic impact including adverse pregnancy outcomes, atherosclerosis, rheumatoid arthritis, diabetes, and aspiration pneumonia. The resulting chronic inflammation, periodontal disease, affects the majority (50%–90%) of the worldwide population. Pathogenic gram-negative anaerobic bacteria populating the tooth-associated biofilm may cause an inflammatory reaction (periodontitis). 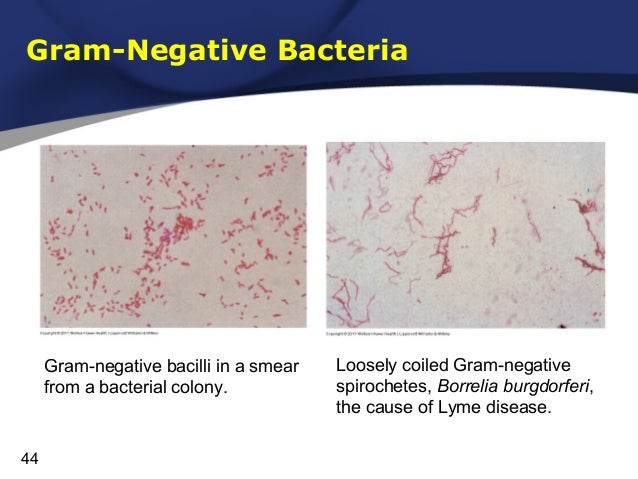 
Hans Helmut Niller, Janos Minarovits, in Epigenetics in Human Disease (Second Edition), 2018 21.2.1 Epigenetic Alterations Induced by Bacterial Infections in Periodontal Disease
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
